
The heyday of cocaine in our nation’s history was arguably the late 1970s through the early 1980s. In the summer of 1980 Richard Pryor set himself on fire while freebasing cocaine, an incident which Pryor mined for a memorable bit in the 1982 movie Richard Pryor: Live on the Sunset Strip. The 1981 movie Modern Problems featured a fantasy sequence in which the protagonist, played by Chevy Chase, exploits his telekinetic powers to vacuum up a roomful of coke, which also brings us, inevitably, to Brian De Palma’s Scarface, which is probably the ultimate cokehead masterpiece in American history. Obviously Woody Allen featured an iconic coke gag in the 1977 classic Annie Hall when he sneezed into a friend’s coke stash.
At the time, there was considerable sentiment around the country that after marijuana, cocaine might be the next drug to “go mainstream.” It was even considered non-addictive! In retrospect, this was never in the cards, however, many people thought it was on the cusp of becoming societally acceptable.

One of the signifiers of the time were underground “manuals” to the coke life. Since the drug was and is illegal, there was a shortage of authoritative guides to the drug and its chemistry, paraphernalia, and lifestyle accoutrements, and intrepid authors willing to make a fast buck tried their hardest to fill in the gap. We’ve already covered The Gourmet Cokebook: A Complete Guide to Cocaine, which dates from 1972. The subject today, however, is the 1976 guide The Cocaine Consumer’s Handbook by David Lee and his 1981 follow-up/expansion, The Cocaine Handbook: An Essential Reference.
These books are difficult to find today, and they fetch high prices on the collector’s market. And there’s not a lot of information about who David Lee is or was. The two books are much heavier on chemistry than, say, what kind of coke spoon goes with your style of shag rug. They provided useful information about the sources of cocaine and the sequence of events that starts with someone harvesting from the coca leaf, most likely in South America, and ends with a rolled-up piece of legal tender being placed in a user’s nostril. Lee described what happens at each stage, as the product moves from cook to alchemist to dealer to user, and also offered information the laws for all 50 states as well as the location of testing labs and treatment centers.

Lee was explicitly “anti-drug” in that he was not an activist pushing for legalization and his guide was mostly meant to increase the awareness of how to test for safe or pure cocaine. Lee described how samples are tested for common adulterants and impurities. His preferred method for testing involved putting the cocaine into Clorox, and if you scour the Internet you can find enough derisive references to it that one can safely categorize the conclusion as “controversial” if not totally debunked (again, depends on whom you ask). One disgruntled reader went so far as to describe Lee as a “shill for Clorox,” which seems a bit unlikely.
What follows are scans of some of the pages from the two books as well as a glossary taken from the longer second book, The Cocaine Handbook, which has been turned into HTML format if you want to experience that. As stated, it’ll run you hundreds of dollars if you want to have a copy on your coffee table (to cut lines on).

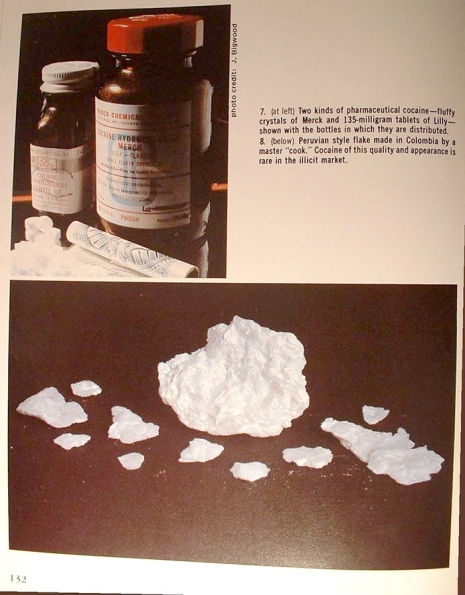





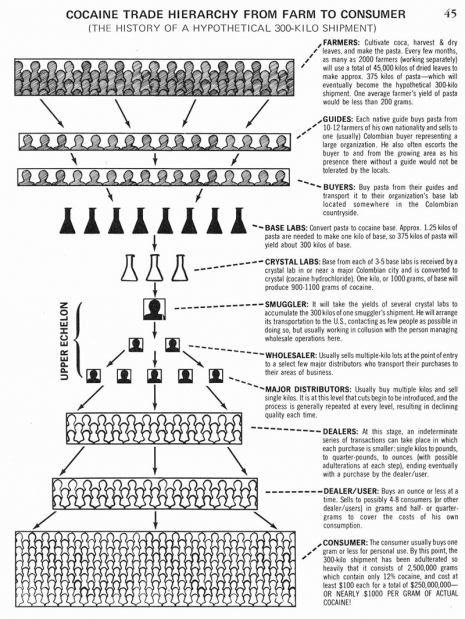
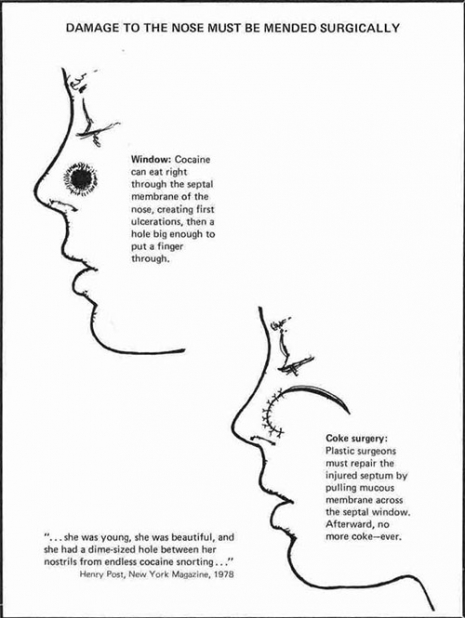


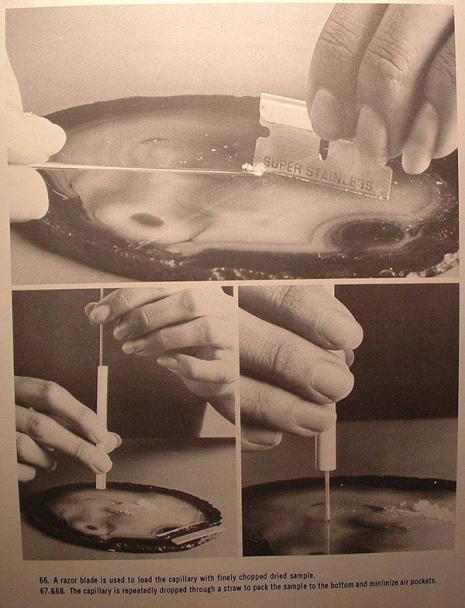
Note the exaggerated length of the pinky fingernail in this one.
Glossary [keep in mind this lingo is more than 35 years old]:
absolute (alcohol): 100% (alcohol), anhydrous, having no trace of water in itacetone: a flammable organic solvent often used as a degreasing cleaner—it mixes with water in all proportions
adulterant: a substance added to a sample to decrease the concentration of the original material
agua de coca: water infusion of coca leaves, drunk as a tea in South America
alkaloid-a nitrogen: containing organic com¬pound which reacts like a base, examples are caffeine and cocaine
ammonium hydroxide: the basic compound which results when ammonia gas reacts with water—it is the active ingredient in household ammonia cleaner
amphetamine: a synthetic central nervous sys¬tem stimulant—the word “speed” and amphet¬amine are almost synonymous
base smoking: the heating of cocaine base to vaporize it and inhaling the vapors—usually done in a glass pipe
baseable: the property of a compound which will make it soluble in an organic solvent like petroleum ether when the pH is made basic benzocaine—a synthetic local anesthetic with a melting point of 88-92°C
benzoic acid: an organic acid which is one of the building blocks of the cocaine molecule
benzoylecgonine: a combination of benzoic acid and ecgonine—each is a building block of the cocaine molecule
Bolivian style cocaine hydrochloride: also known as “Bolivian rock” a dense crystal-like form of illicit cocaine hydrochloride—the origin is not necessarily Bolivia
butacaine: a synthetic local anesthetic, the sulfate has a melting point of 138.5-139.5°C
caffeine: an alkaloid which is a stimulant and found in coffee, tea and numerous other plants (mp 238°C)
capillary tube: when referring to melting point tests, it is the thin-walled tiny glass tube which is filled with powdered sample and observed while heating
Cheyne-Stokes respiration: a pattern whereby breathing builds deeper and deeper to a point, then becomes progressively shallower, with a period of no breathing between cycles cinnamylcocaine: an alkaloid related to cocaine where cinnamic acid replaces benzoic acid in the molecule
Clorox test: a method used to test cocaine for adulterants which uses a glass of liquid laundry bleach (5.25% sodium hypochlorite)
coca: a shrub originating in the Andes from which the alkaloid cocaine is derived
cocaine: the alkaloid obtained from coca leaves which has activity both as a local anesthetic and a stimulant of the central nervous system cocaine base: the form the cocaine molecule has in a basic solution having no electrical charge; it is soluble in organic solvents
cocaine hydrochloride (pharmaceutical): a salt form of pure cocaine which has an electrical charge and is soluble in water
cocamine (truxillines): a group of alkaloids related to cocaine in structure but not having the pharmacological activity of cocaine
crystal: for cocaine, refers to cocaine hydrochloride
crystal lab: an illicit lab, usually in South America, which converts cocaine base into cocaine hydrochloride
cut: slang for adulterant
DEA: the acronym for Drug Enforcement Administration
duff: term used to describe the fine powder which passes through a screen when a sample is gently shaken
ecgonine: the central molecular building block of the major coca alkaloids, from the Greek for son or descendant
empanada: the form the crystals of cocaine hydrochloride take after drying inside the flattened filter paper, the appearance being similar to a South American meat pie
Erythroxylum coca: one of the species of coca Erythroxylum novogranatense: one of the species of coca
Erythroxylum truxillense: one of the varieties of coca
ether (diethyl): the solvent traditionally used to make cocaine hydrochloride; this ether is extremely flammable and forms explosive peroxides on exposure to air
flake cocaine: a form of cocaine hydrochloride that appears to be made of many paper thin layers of pearly crystals, sometimes “Peruvian flake”; it is more properly Peruvian style flake since it refers to the form rather than the country of origin
foil burn test: a test for cocaine hydrochloride adulterants where a small sample is heated from below on a sheet of aluminum foil
forensic (labs): refers to an argument used in court, as a lab it usually means a police lab
freebase: the slang term for the alkaline form of the cocaine molecule (not the salt form) which is used for smoking
gas chromatograph(y): a usually analytic technique which separates a mixture of compounds from each other in a stream of moving gas which passes through a specially designed tube
Huanaco: a region in South America where a species of coca is thought to originate
hygrines: a group of coca alkaloids which are oily bases
inositol: a type of sugar-alcohol (mp 225-227°C) which is frequently used to adulterate cocaine hydrochloride
Ipadu (or Amazonian) coca: one of the species of coca
lactose: a sugar also known as milk-sugar (mp 201-202°C) which is sometimes used to adulterate cocaine hydrochloride
lidocaine: a local anesthetic (mp 68-69°C) frequently used to adulterate cocaine hydrochloride or sold as cocaine hydrochloride itself
mannitol: a sugar-alcohol (mp 166-168°C) which is currently the most commonly seen adulterant of cocaine hydrochloride
mass spectrometer (high resolution): an analytical tool which fragments and separates molecules into various building blocks which can frequently provide enough information to reconstruct what the original molecule looked like
melting point: the temperature or range of temperature at which a solid turns to a liquid melting point test—the determination of the melting point or range of a sample to estimate its degree of purity; impurities lower the melting point and widen the range of the melting point
methanol: methyl alcohol, also known as wood alcohol, a poisonous flammable solvent methyl benzoate: a compound with a strong wintergreen aroma; it is produced when cocaine is heated with absolute methanol in a basic solution
mother-of-pearl cocaine: cocaine hydrochloride crystals in flake form which have the appearance of mother-of-pearl
non-baseable: the chemical characteristic of an adulterant which leaves it in the water phase when the cocaine base is filtered or extracted with organic solvent
pasta: cocaine sulfate (with other alkaloid sulfates), a water soluble salt produced in an early stage of refining illicit cocaine
pasta lab: the lab, usually in South America, where coca leaves are brought to be turned into pasta
Peruvian style cocaine hydrochloride: a flake form of cocaine hydrochloride crystals which in the past was thought to originate In Peru; now it refers to the form of crystal; the alkaloid can come from anywhere
petroleum ether: not a true ether but a fraction of petroleum distillation, similar to gasoline in chemistry
pH: a scale used to measure acidity, pH 1 is extremely acidic, pH 14 extremely basic, and pH 7 is neutral
phenylpropanolamine: a compound (mp 101-101.5°C) used in antihistamines and non-prescription diet pills, similar to amphetamines in chemistry and sometimes used to adulterate cocainepotassium permanganate: a chemical which is frequently used to oxidize and break down or¬ganic compounds; in refining cocaine it can be used to eliminate non-cocaine alkaloids
Potpourri: a mythical country or region where many samples of illicit cocaine really originate; most cocaine now results from leaves gathered from diverse regions
procaine: a local and spinal anesthetic (mp 61°C) sometimes used to adulterate cocaine
quinine: an alkaloid from cinchona bark usually used to treat malaria; the sulfate is sometimes used to adulterate cocaine, and is dangerous to the user
rock cocaine: a form of cocaine hydrochloride which is dense, unlayered, and similar in appearance to rock salt
snorting: taking a drug by inhalation through the nose
spectrophotometer: an optical instrument which reads how much light of a particular wavelength a sample absorbs
tetracaine: a local anesthetic (mp of hydrochloride, 147-150°C) sometimes used to adul¬terate cocaine
Thiele-Dennis tube: a special glass tube used in determination of melting points; it allows slow, even elevation of temperature
thin-layer chromatography: usually analytical method for separating a mixture into its pure compounds; it uses a thin, solid film on a plate and a special solvent which slowly moves across the plate and moves the different compounds different distances
tropacocaine: one of the alkaloids in coca which is related to cocaine
truxfllic acid: one of the building blocks of alkaloids related to cocaine; they take the place of benzoic acid in cocaine
wash: the process of mixing a solid and solvent which will not dissolve the solid and separating the liquid and solid by decantation or filtration; impurities soluble in the liquid are removed in the process
Previously on Dangerous Minds:
‘The Gourmet Cokebook: A Complete Guide to Cocaine,’ 1972